News Raw Materials
An update on Korea Reach
From January 1st 2019, the revised Korea REACH, the Act on the Registration and Evaluation of Chemicals, has been implemented. What does this mean for the coatings industry?

An update on Korea Reach. Image source: andriano_cz _StockAdobe.com
By Bryan Zhou, CIRS
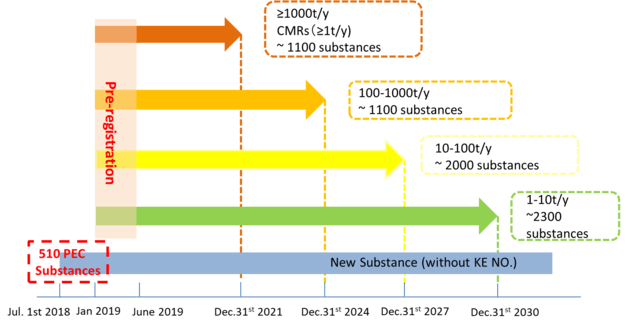
The deadline of pre-registration was passed on June 30th 2019, there have been more than 177,000 pre-registrations submitted for more than 16,000 substances. If non-Korean companies did not complete their pre-registration before and still plan to export their chemical products to Korea, there is the option to carry out late pre-registration.
Non-Korean manufacturers or formulators can process late pre-registration by appointing a Korean Only Representative. The condition of late pre-registration is the average tonnage band exported to Korea from 2016 to 2019 is less than 1ton/year. Documentation will be required by the authorities to prove that the annual tonnage band from 2016 to 2019 was below this threshold.
Currently most registrants are in the process of joining the CICO (Chemical Information Communicative Organization) and participating in CICO activities, as the deadline for the pre-registration of the >1000 and the 100 – 1000 tonnage band fast approaching (Fig.1). CICO is similar to SIEF under EU REACH and the only way to join the CICO is by completing (late) pre-registration.
CICO activities include CICO Role decision, LR election, Consortium formation, data sharing discussion, LOA reimbursement mechanism discussion, etc. Under usual circumstances the authorities require the Consortium meetings to be carried out in person. However, due to COVID-19 pandemic, online meetings are now being held in their place. The formal procedure for the registration of existing chemicals is similar as EU REACH with joint submissions being mandatory but there is also an option to Opt-out under K-REACH.
In all CICO activities, the main issue which many registrants have is how to decide their role in the CICO. There are four different roles: Lead Registrant, Active Member, Passive Member and Dormant. Role decision is very important to all registrants and many factors shall be considered when making the decision, including: deadline, prior-period investment of time and budget, qualified existing data and reports, etc. What are the specifics of each of the four roles?
Lead Registrant (LR)
The lead registrant will lead the formation of the consortium, the work of the consortium and the joint submission of substances. Usually, the LR is involved in the decisions surrounding the data selection and data sharing principle as well as maintaining data ownership and registration initiative, which means LR will take the initiative to complete K-REACH registration and enjoy a reimbursement of the registration fee. The LR may also enjoy lower cost or even benefit from the registration.
Active Member (AM)
Active members will participate in the formation and work of the consortium as being a member of the consortium. An AM is involved in the decisions surrounding the data selection and data sharing principle as well as maintaining data ownership, registration initiative. An AM can obtain a reimbursement of the registration fee and a lower cost.
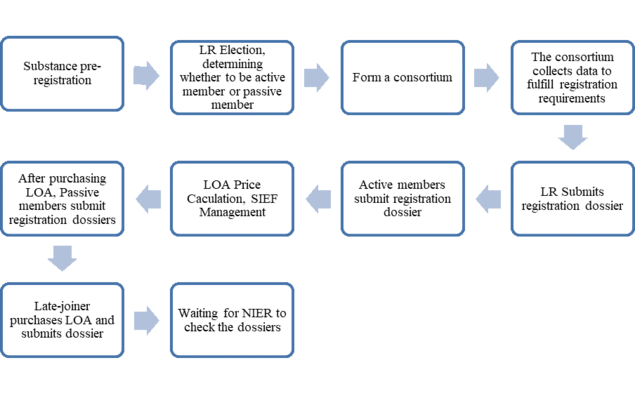
Passive members (PM)
Passive members will participate in the relevant work for a joint submission without joining the consortium. They are not required to bear the registration expenses in the early stages and can purchase the LOA from the consortium to participate in the joint submission. The registration fee is higher than the lead registrants and active members. PM will not have LOA reimbursement.
Dormant
A dormant does not participate in the relevant work of joint submission and will not join the consortium. It does not need to bear the registration expenses in the early stages. Dormant has no decision rights, nor data ownership. The registration fee for dormant is higher than the lead registrants and active members.






